Sepsis & Endotoxin (LPS)
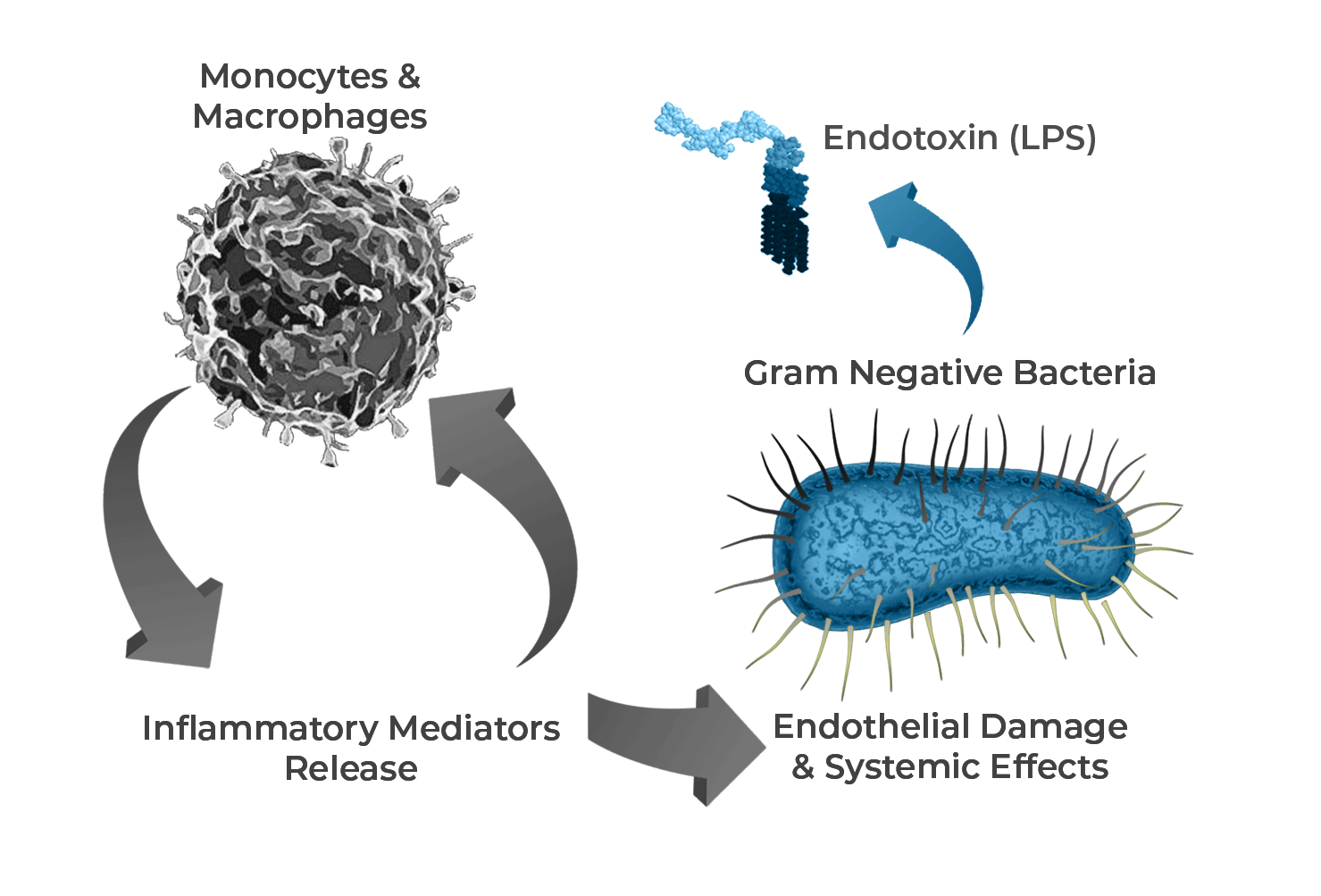
Endotoxin, also referred to as Lipopolysaccharide (LPS), a component of the cell wall of Gram negative bacteria, is one of the main triggers of the pathogenesis of septic shock and multiple organ failure. Its entry into the bloodstream stimulates monocytes/macrophages that, once activated, produce and release cytokines such as TNF and IL-6, nitric oxide and other mediators that induce systemic inflammation, endothelial damage, hypotension (shock), and multiple organ dysfunction.

The EAA™ is a chemiluminescent bio-assay based on the oxidative burst reaction of activated neutrophils to complement coated LPS-IgM immune complexes. The IgM antibody, a key reagent, is specific for the Lipid A portion of endotoxin (LPS). In the presence of LPS, the ensuing oxidative burst results in light emission in the presence of luminol. Each EAA™ test relies on 3 reactions: the first reaction is a negative control which allows each patient to be their own control, the second is the test sample and the third is a maximum chemiluminescence calibrator with a high level of exogenous endotoxin. The maximum calibrator is necessary as endotoxins from different species of Gram negative bacteria have a slightly different reactivity in the EAA™. E. coli O55:B5 is used as the standard as it has a mid-level reactivity in the assay.

Ionic: guides the formation of the link/bond;
Hydrophobic: breaks the spatial order of the acyl chains of LPS and neutralizes the toxicity, transforming LPS and Polymyxin-B into a single unimolecular compound.
PMX (Toraymyxin PMX-20R) removes endotoxin from the blood using the electrochemical properties of Polymyxin-B, while avoiding known side effects of its systemic administration. The PMX cartridge contains polystyrene composite woven fibre with Polymyxin-B immobilized on the surface through covalent bonding. The blood flows radially and uniformly through the fibres at a rate of 80-120 mL/min before exiting the cartridge. The recommended dosing of hemoperfusion is two 2-hour hemoperfusion events in a 24 hour period. The in-vitro adsorption capacity (human blood) was measured as 20 μg (Romaschin et al., 2017).

